Optimizing clinical trials: Getting the right results by keeping it real
We surveyed clinical research leaders across biopharma companies and contract research organizations (CROs) to learn how they've used real-world data to solve persistent challenges to successful clinical trials.
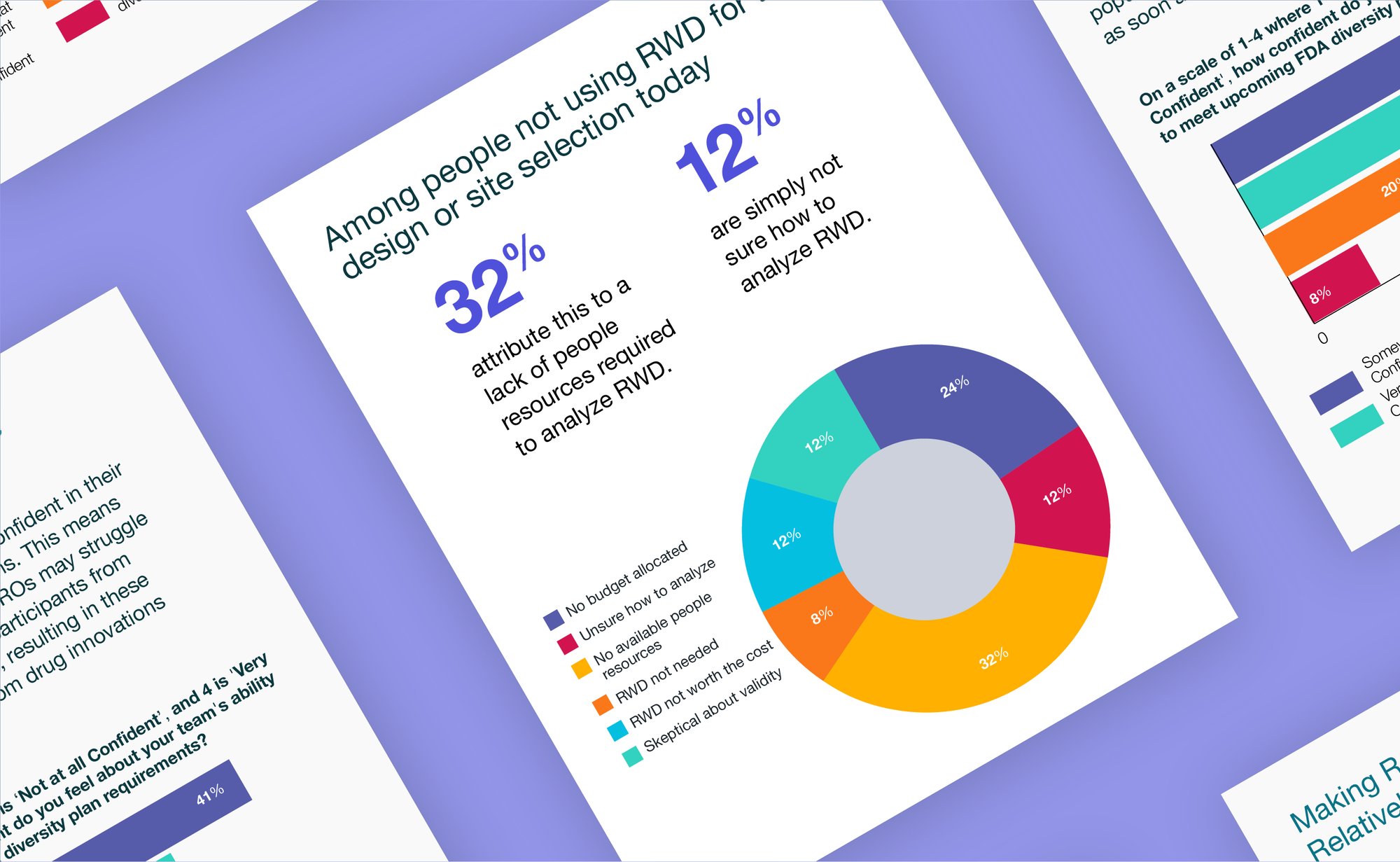
Despite years of industry-wide efforts to tackle challenges related to protocol optimization, patient identification, and site selection with a mix of different tactics, we still see underperforming trials. What tactic hasn’t been utilized to its full potential? Real-world data (RWD).
Download "Optimizing Clinical Trials: Getting the Right Results by Keeping it Real" and discover how the top biopharma companies and CROs are leveraging RWD to transform clinical trials.